Nuclear medicine is a branch of medicine and medical imaging that uses the nuclear properties of matter in diagnosis and therapy. More specifically, nuclear medicine is a part of molecular imaging because it produces images that reflect biological processes that take place at the cellular and subcellular level. The following article gives a peek at the diagnostic role of Nuclear Medicine.
Nuclear medicine has come a long way since the first imaging days of the rectilinear scanner of Benedict Cassen and Anger Camera of Hal Anger (who passed away on 31 October 2005) in 1950s. Particularly over the last couple of decades the progress line took a more upward angle. The young scientist at Neils Bohr institute, when he first used radioisotope to expose recycling of leftover mashed potatoes by his landlord (who was supplying his food), would have little expected his innovative method to become a complete specialty by itself in medical science one day.
The progress in nuclear medicine can be largely classified into diagnostic and therapeutic divisions. The diagnostic division can further be viewed in three different areas, namely- imaging technology, radiopharmaceutical development and molecular imaging.
Since the analog imaging gave way to digitisation, the imaging technology started growing in leaps and bounds and it is virtually the rejuvenation and metamorphosis of ‘unclear medicine’ in clinical application into a ‘new clear medicine’. While faster electronics and more efficient electronic collimation optimised the data conversion and drastically cut down the imaging time, the development of faster crystals contributed significantly. Though Positron Emission Tomography has been in evolution for about 30 years, only in the last decade did PET imaging take off to conquer the wider scope of clinical applications. Thanks to the availability of baby cyclotrons (relatively low energy proton accelerators) and the tireless campaign by the group- Institute of Clinical PET (ICP), which later transformed into Academy of Molecular Imaging (AMI), who accumulated and provided all the required data to satisfy the decision makers and are conscious of evidence based medicine, for reimbursements. Now with the arrival of hybrid imaging in the form of PET�CT and SPECT-CT, a whole new horizon is wide open in diagnostic nuclear medicine. Virtually all new scanners sold in the last couple of years by all three big manufacturers (GE, Philips and Siemens) are hybrid scanners, thus making standalone PET a thing of the past. Recent development of faster crystals like LSO (Lutetium Ortho-silicate by Siemens and LySo by Philips) along with faster electronics has enabled to cut the PET imaging from about 40 minutes a patient (7 minutes a bed position) to less than 10 minutes (90 seconds a bed position). This means increase in patient throughput, which can make it more viable commercially. The ‘time of flight’ technology, which calculates the distance traveled by each photon to discriminate the coincidence events from random photons requires timing the events with nano second precision. This finally took shape, thanks to the faster computers. Commercial scanners have recently been marketed by Philips.
In the field of radiopharmaceuticals, the evolution of FDG (2 fluro 5 deoxy glucose) as PET imaging agent has changed oncologic imaging forever. Today FDG is the most commonly used PET tracer and it is not going to change in the near future. Development of Rubidium generators has set a new standard in myocardial perfusion imaging, though the usage is still limited to a handful of centres, even in developed countries. F18 DOPA, F18 FDDNP, F18 FLT and G68 DOTA among others are available commercially and are in clinical use at many leading nuclear medicine centres.
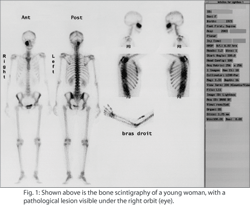
Disease starts at the cellular level. Metabolic and physiological changes precede the anatomical changes, which happen eventually and can be detected by conventional imaging modalities like ultrasound, CT and MRI. Some diseases may not even cause anatomic changes. Molecular imaging aims to detect the disease process before it causes anatomic changes.
More than a score of F18 and C11 based agents are at various stages of clinical trials and in the next few years several are expected to be available commercially. Gamma imaging field has also seen the arrival of many new and
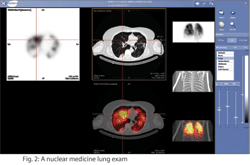 improvised tracers, most of them technetium – based. Use of non Tc based tracers is slowly dwindling, except for Iodine which continues to enjoy popularity. In addition to I131, which retained support as thyroid imaging agent, newfound interest in I124 as positron emitting isotope may result in some landmark radiopharmaceuticals in coming years due to its favorable chemistry.
improvised tracers, most of them technetium – based. Use of non Tc based tracers is slowly dwindling, except for Iodine which continues to enjoy popularity. In addition to I131, which retained support as thyroid imaging agent, newfound interest in I124 as positron emitting isotope may result in some landmark radiopharmaceuticals in coming years due to its favorable chemistry.
Every few years nuclear medicine faces a threat from other imaging modalities and some how or other emerges with a renewed vigor. First it was CT virtually wiping out brain and liver, spleen SPECT. Nuclear imaging rebounded with myocardial perfusion scintigraphy, which in turn recently came under threat from CT angiography. Radio Nuclide Ventriculography, the gold standard for left ventricular ejection faction, has already been dominated by echoca-rdiography for various reasons. With emergence of PET and hybrid imaging, nuclear medicine is enjoying a stint of rejuvenation currently. But an exciting future lies ahead in molecular imaging!
We all know that the disease process starts at cellular level. Metabolic and physiologic changes precede the anatomical changes, which happen eventually and can be detected by conventional imaging modalities like ultrasound, CT and MRI. Some diseases may not even cause anatomic changes. Molecular imaging aims to detect the disease process before it causes anatomic changes. Most top-notch nuclear medicine centres have strong research programs at various stages developing various molecular probes. In addition every pharmaceutical corporation worth its name has a research wing with micro PET and animal imaging. With all these happening, nuclear medicine is not just looking forward to a great future but actively working towards a concrete goal, that of finding the root cause of disease at molecular level and working towards prevention – thus working for a disease free society.
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter , Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!










