Sanofi Healthcare India Pvt. Ltd. today announced that it has received marketing authorisation for Dupixent® (dupilumab), the first biologic medicine for the treatment of moderate-to-severe atopic dermatitis in adults whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. Dupixent® can be used along with or without topical therapy.
Globally, Dupixent® has transformed the treatment landscape for patients around the world by targeting the type 2 inflammation that underlies the disease, rather than broadly suppressing the immune system.
Anil Raina, General Manager, Sanofi Specialty Care (India) stated, “Dupixent® receiving marketing authorisation in India is a significant milestone, as we now have the opportunity to offer our first-in-class and best-in-class therapy to treat people living with atopic dermatitis, in India. Approved in the U.S., the European Union, Japan, and more than 60 countries for one or more indications other than atopic dermatitis, Dupixent® is the first and only biologic medicine in India that has shown significantly improved disease signs, symptoms, and quality of life measures, for this particularly difficult-to-treat skin condition.”
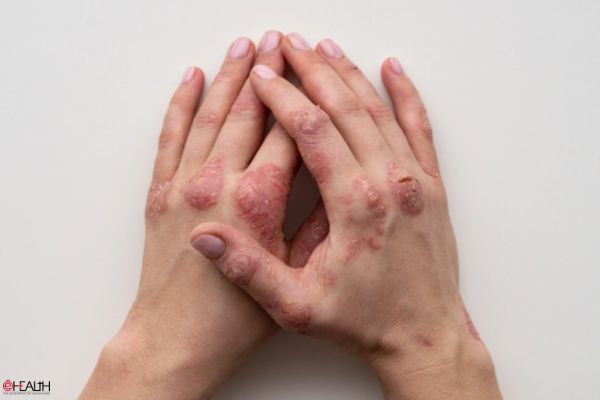
Atopic dermatitis, a form of eczema, is a chronic type 2 inflammatory disease with symptoms often appearing as a rash on the skin. Moderate-to-severe atopic dermatitis is characterised by rashes often covering much of the body and can include intense, persistent itching and skin dryness, cracking, redness, crusting, and oozing.
Dr Shalini Menon, Country Medical Lead, Sanofi (India) said, “The prevalence of atopic dermatitis in adults in India ranges from 2% to 8% and there has been a trend of increasing prevalence observed in India. Many often struggle to control their disease with the treatment options currently available. Itching is one of the most burdensome symptoms for patients and can be debilitating. People living with moderate-to-severe atopic dermatitis can experience unbearable symptoms and have significantly impaired quality of life, including disrupted sleep, and increased anxiety and depression symptoms. The chronicity and often visible lesions lead to considerable social stigma. Through studies, Dupixent® has shown that it helps clear the skin, manage the persistent debilitating itch, and improve overall quality of life along with proven long-term safety.”
With more than 600,000 patients being treated with Dupixent® globally, Dupixent® will soon be available as an option for controlling moderate to severe atopic dermatitis for adults in India. Dupilumab is being jointly developed by Sanofi and Regeneron under a global collaboration agreement.
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter , Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!



















