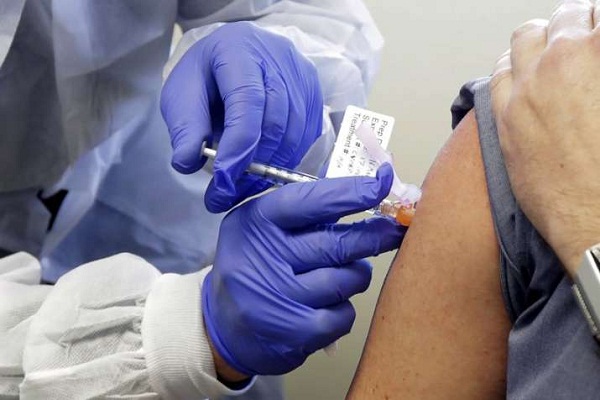
Zydus Cadila has started human clinical trials of its COVID-19 vaccine candidate ZyCoV-D, the drug firm announced. The company will be enrolling over 1,000 subjects across multiple clinical study sites in India during trials, it said in a regulatory filing on Wednesday.
The adaptive phase I/ II human clinical trials of ZyCoV-D have commenced with the first human dosing, it said, adding that “the adaptive phase I/II dose escalation, multi-centric study will assess the safety, tolerability and immunogenicity of the vaccine.”
Also read: DCGI nod to Zydus Cadila for world’s first drug to treat liver disease
Earlier this month, Zydus had received approval from domestic authorities to start human trials for its COVID-19 vaccine contender – the second Indian pharmaceutical firm to get such nod amid a surge in novel coronavirus infections worldwide.
It got approval a few days after India’s first indigenous COVID-19 vaccine candidate COVAXIN, developed by city-based Bharat Biotech in collaboration with Indian Council of Medical Research and National Institute of Virology, got the nod for human clinical trials from the Drug Controller General of India.
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter , Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!




















