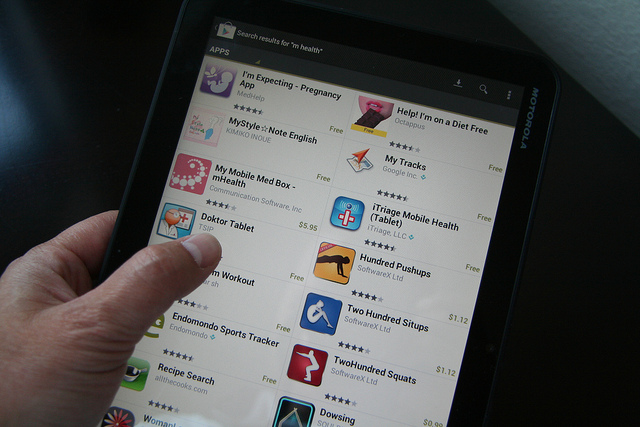
There was a proposal introduced by the Food and Drug Administration that would allow it to regulate smartphone and tablet apps that relate to health and medical needs. The FDA seeks input on its proposed oversight approach for certain mobile applications specific to medicine or healthcare called mobile medical applications (‘apps’) that are designed for use on smartphones and other mobile computing devices. This would encourage the development of new apps, focuses only on a select group of applications and will not regulate the sale or general consumer use of smartphones or tablets. The mobile applications the FDA is eyeing for regulation include those that “are used as an accessory to a medical device already regulated by the FDA,” such as apps that allows doctors to make a specific diagnosis by viewing a medical image on a smartphone or tablet. This would further transform a mobile communications device into a regulated medical device by using attachments, sensors or other devices. Also included would be apps that “such as those that can turn a smartphone “into an ECG machine to detect abnormal heart rhythms or determine if a patient is experiencing a heart attack. The FDA is looking for the public to comment and weigh in on its app regulation proposals.

Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter , Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!