
Dr Chirag Trivedi, President, Indian Society for Clinical Research (ISCR) elucidates to Prathiba Raju about the challenges of fast tracking vaccines and why it is critical to evaluate the safety and efficacy data generated at each stage.
How much time does it take it to develop a vaccine? Why each stage of the clinical trial is crucial and why should we adhere to the same? What are the potential options to fast track them shortcuts?
Generally, it takes around 10 to 15 years for a vaccine to be developed and be made available for being administered to the general public. As in the case of drugs, even vaccines have to undergo the various phases of development:
The first is the pre-clinical phase where it is tested in animals – this may take 1-2 years.
Next is the Clinical Trial Phase 1: The main aim of this is to find the safety and immunogenicity potential of the candidate vaccine. This is done usually in 20-80 volunteers and may take 1-2 years. Only after the results are positive, the candidate progresses to the next phase of clinical trial i.e., Phase 2.
Clinical Trial Phase 2: The main aims are to evaluate the safety, immunogenicity potential, doses, immunization schedules, etc. This is conducted usually in a few hundred people and it may take 2-3 years for this phase. The results of this will take the candidate vaccine to the next pivotal phase i.e., Phase 3.
Also read: India’s tryst with Covid-19 vaccine
Clinical Trial Phase 3: The main aim of this vaccine is to evaluate the safety and efficacy of the candidate vaccine in a large population covering the population it is intended for. This phase is done in thousands of people and it may take 4-5 years or even longer depending upon the disease being studied.
After the success of the Phase 3 clinical trial, the entire dossier is submitted to the drug regulatory authorities of the individual countries, who will thoroughly evaluate it and then decide to grant the marketing authorization for it to be administered to the people in that country.
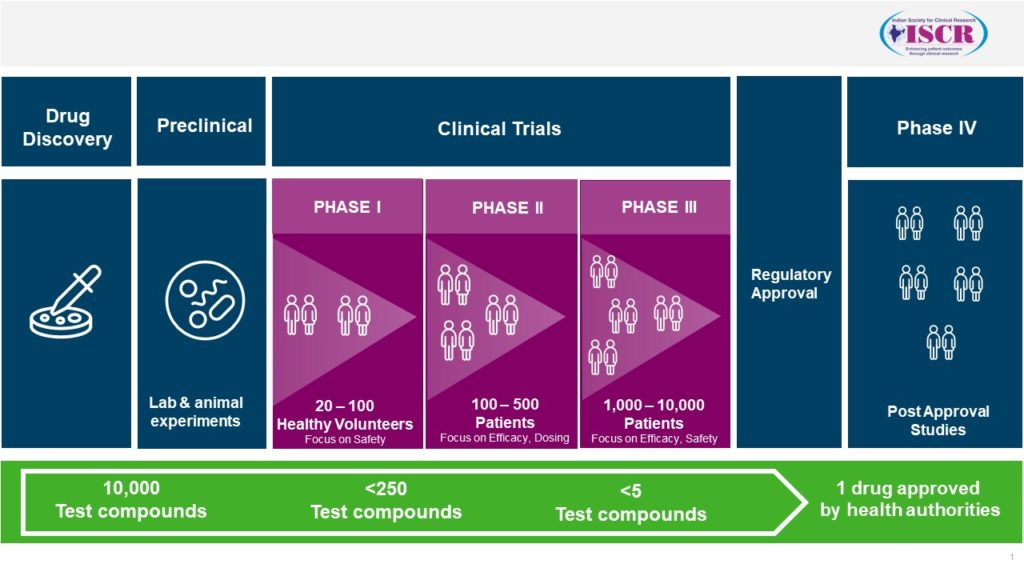
Phase 4: Evaluating the safety and effectiveness of the vaccine in large scale studies after it has been marketed.
Can you brief us about how fast-tracking is done as per globally accepted norms. Is it possible to develop a safe vaccine within those norms?
Since the COVID-19 pandemic has become a dreaded disease that has caused havoc across the world, it is imperative for all stakeholders, for e.g., researchers, bio-pharmaceutical companies, regulators, governments, medical institutes and the site teams, ethics committees, etc., to work with unprecedented speed. This is being currently observed in the vaccines that are developed for this disease by various organizations across the world. Some steps being taken to significantly shorten these timelines are: (a) working with top speed to develop clinical trial protocols based on the existing as well as evolving disease knowledge (b) The regulators and the ethics committees have significantly shortened their approval timelines – from a few months earlier to being approved in a few days during this COVID-19 pandemic, without any compromise in the quality of their review. (c) Combining Phase 1 and Phase 2 trials by including hundreds of individuals in these studies and using adaptive designs. (d) Maintaining top speed in collecting, cleaning, analyzing and reporting data.
Having said this it is pertinent to note here that while speed is definitely the need of the hour, it is important that all the applicable guidelines and regulations are followed while planning, conducting and analyzing these studies.
Vaccine development takes about 10 to 15 years. What are the risks involved in compressing the traditional timeline? Can you provide your thoughts both on the aspect of technological innovation and the monetary risk?
Various challenges exist in developing vaccines. One of it is that the organisms may keep mutating. Hence, it is important for us to make sure that sufficient immunogenicity is generated against these organisms. Also, since the timelines are compressed, it is important to thoroughly and critically evaluate the safety and efficacy data generated at each stage. One also needs to be watchful if the use of this vaccine leads to vaccine-induced enhancement wherein it may make the disease worse if a vaccinated person gets infected by the virus.
Various companies are trying new versus the established platforms for developing candidate vaccines. The newer platforms need to be critically evaluated.
Traditionally, the large commercial scale manufacturing of vaccines start once these trials are successful. This adds further to the timelines for vaccine availability for the general public. During these times of the COVID-19 pandemic, various vaccine manufacturers have already started, though at risk, vaccine production on a large commercial scale. This will help in significantly shortening the vaccine availability time if the Phase 3 trial results are successful. Thus, the companies are taking a commercial risk of starting the large scale production of these vaccines even before the clinical trial results are out.
For any drug approval, there’s always a balance between benefit and risk, if at all the vaccine is developed in a short span of time should it be used to people who are exceptionally at risk, based on expected benefit, or should they wait for proven benefit?
If the vaccine candidate’s clinical trials are successful, it can be approved by the country’s government and regulators for an emergency use by authorizing it to be used for people who are on the frontline in fighting this disease for e.g., the healthcare workers, people providing essential services, etc. Large scale data will be required to see the long term safety and effectiveness of these vaccines. This will be possible when the vaccines will be administered to millions and billions of people across the globe after they are made available.
As the history of drug development has shown disappointments, even in the final-stage trials. Is the fast-tracking mode safe what will be the consequences?
Yes, the drug and vaccine development is a long, costly and risky process with chances of failure at different stages of clinical trials.
Having said this, the need for having a vaccine against this dreaded pandemic is more today than ever before. Hence, we all have to work with top speed in developing and testing vaccine candidates. But, while working with this lightning speed, it is important for us to follow all the stipulated guidelines and regulations pertaining to clinical trials.
People world over are eagerly waiting for a drug and vaccine against this dreaded disease. Thus, speed, quality and ethics should co-exist. We have always adhered to this and hence, it should be the case today and in the future too!
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter , Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!




















