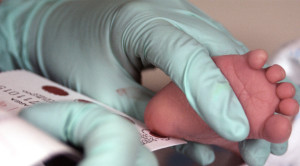
Trivitron Healthcare has launched and patented NeoMass AAAC Kit (CE Approved) for expanded panel of Newborn Screening in India. This kit can detect up to 50 disorders that a newborn baby might be born with or without any immediate symptoms or visible manifestations, making it the broadest available panel of biomarkers.
Dr GSK Velu, Chairman and Managing Director of Trivitron Group of Companies said, Trivitron Healthcare is one of the largest provider for Newborn screening solutions in the world and our new patented NeoMass AAAC kit is the first kit in the world to detect up to 50 disorder. Trivitron Healthcare is always at the forefront of developing affordable technologies for Indian market.
The company aims to create awareness among the target audience about Newborn screening and its alarming need to make it mandatory in India. The testing process involves pricking the heel of a newborn baby to collect a few drops of blood on a filter paper within 72 hours of birth and test for detection of life threatening disorders. The NeoMass AAAC kit would lead to screening of inborn metabolic disorders based on a technology called tandem mass spectrometry which allows fast and accurate determination of metabolic disorders. The newly patented NeoMass AAAC kit uses tandem mass spectrometry technology.
The recently patented methodology makes it possible to detect deficiencies of all the six enzymes involved in the urea cycle metabolic pathway. The urea cycle is a metabolic pathway occurring in the liver and kidney and aberrant functions of the urea cycle enzymes lead to the buildup of ammonia, a product of protein metabolism. If not treated the outcome is very serious with symptoms ranging from vomiting and increasing lethargy to respiratory distress and even coma and death. Additionally NeoMass AAAC enables an easy and reliable detection of type I tyrosinemia, a serious disease causing dermatologic and neuro developmental problems due to accumulation of tyrosine within the liver and kidney.
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter , Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!